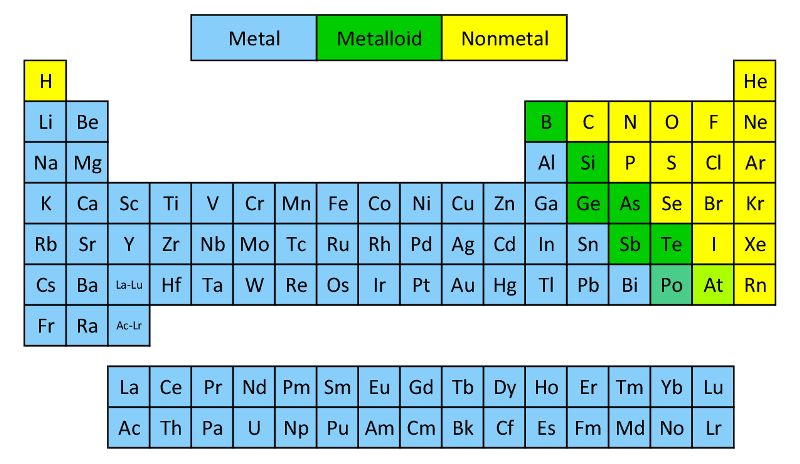
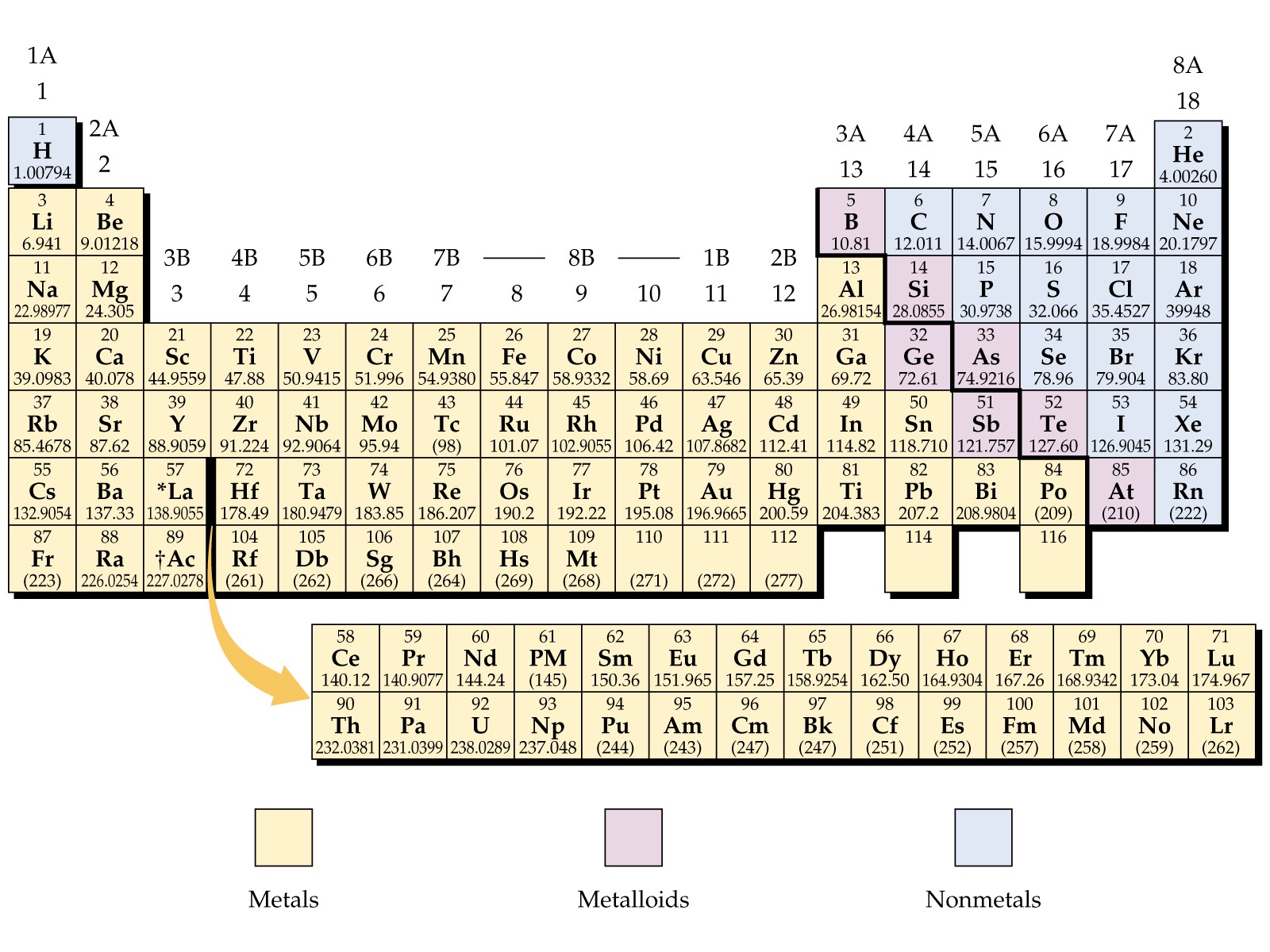
Alkaline-earth metals are Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), and Radium (Ra).Shiny, silvery-white, somewhat reactive metals at standard temperature and pressure.Alkaline-earth metal is a group of six elements are in the Periodic Table.Span from Beryllium (Be) through Radium (Ra).When compounds of alkaline-earth metals are mixed in solutions, the solution become basic or alkaline (having pH higher than 7). Among alkali metals, Sodium is the most abundant.Alkali metals are Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs), and Francium (Fr).Burst into flames when come in contact with water.Stored in oil to prevent reaction with air.Alkali metal is a group of six elements are in the Periodic Table.Spans from Lithium (Li) to Francium (Fr).For example, sodium hydroxide and potassium hydroxide. Alkali MetalsĪlkali metals are named thus because when they react with water they form compounds called alkalies (i.e. A hydrogen atom contains one proton and one electron. Hydrogen is the first element in the Periodic Table. Let’s see how Mendeleev achieved this feat.īefore we begin, there is a small note about the first element. One of the primary reason behind creation of Period Table is to group elements in a way so that all elements with similar properties come in a group. Now let us look at the Periodic Table groups in more details. Groups 9: Lanthanides or Lanthanoid (aka rare-earth elements).Group 4: Post-transition metals (aka other metals).There are a total of ten groups in the Periodic Table, namely: With same number of electrons in the outermost shell the grouped elements behave in similar manner. The column number tells us how many electrons are there in the outermost energy level of element elements in that group. The row number indicates the number of energy levels required by the electrons of elements in that row to fit in.


Elements are placed in Periodic Table in seven rows and 18 columns. All the known chemical elements organized in Periodic Table - first published by Russian chemist Dmitri Mendeleev. So far, a total of 118 elements are known to us.
